First-in-Human Clinical Trials
First-in-Human Solutions for Early-Phase Clinical Trials
With over 30 years of experience and 400 FIH studies completed, we are uniquely positioned to help you achieve critical milestones quickly and reliably. Each year, 50% of our clinical studies are FIH trials.
From protocol design to study start-up and conduct, we translate your nonclinical findings into clear FIH study designs and objectives, ensuring the best chance of finding reliable safety and proof-of-concept data to accelerate your drug development journey.
Need to Conduct a FIH Trial?
Here's Why You Should
Conduct it With Altasciences
With a focus on early-phase drug development, and a proven track record of conducting hundreds of FIH trials, we excel at guiding sponsors through this pivotal stage of development.
- Two Regulatory Pathways—Investigational New Drug (IND) for the U.S.A. and Clinical Trial Application (CTA) for Canada.
- Rapid Study Start-Up enables speedy mobilization to meet your needs. When timelines are critical, we can fast-track start-up to initiate your trials in as little as four weeks.
- Co-Located Clinics and Laboratories provide real-time data access to support faster decisions and adaptive study designs.
- Advanced Data Visualization deliver interactive safety reviews and real-time insights for informed, data-driven decisions.
- A Turnkey solution for First-in-Human Trials to tackle every aspect of your trial from start to finish.
Partner with Altasciences to confidently take your molecule from discovery to first-in-human and beyond. Contact us today to discuss your FIH study requirements!
Two Regulatory Pathways
We specialize in keeping your study on schedule from initiation to completion. With almost 600 beds in three inpatient clinical pharmacology units and a network of several hundred patient sites across North America, we offer dual regulatory pathways—Investigational New Drug (IND) or Clinical Trial Application (CTA).
U.S. IND Pathway
We provide full support, from pre-IND meetings to IND assembly, submission, and maintenance.
CTA Pathway
We handle everything from initial applications to ongoing compliance .
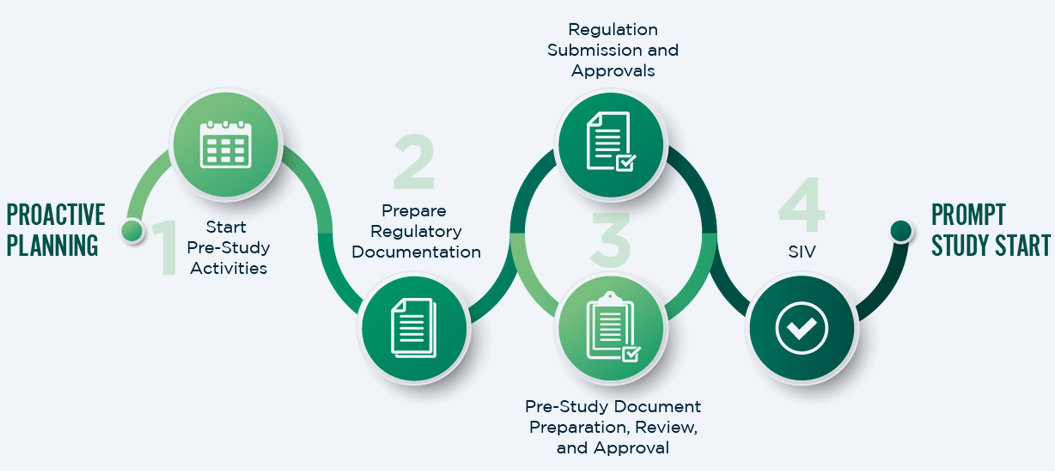
Rapid Study Start-Up
We excel at mobilizing quickly, accelerating start-up timelines to meet your needs. When timelines are critical, we can fast-track start-up and have initiated trials in as little as four weeks. Our teams define key planning elements early to ensure rapid completion of reviews and secure necessary clearances from regulatory authorities, ensuring your clinical activities begin on time.
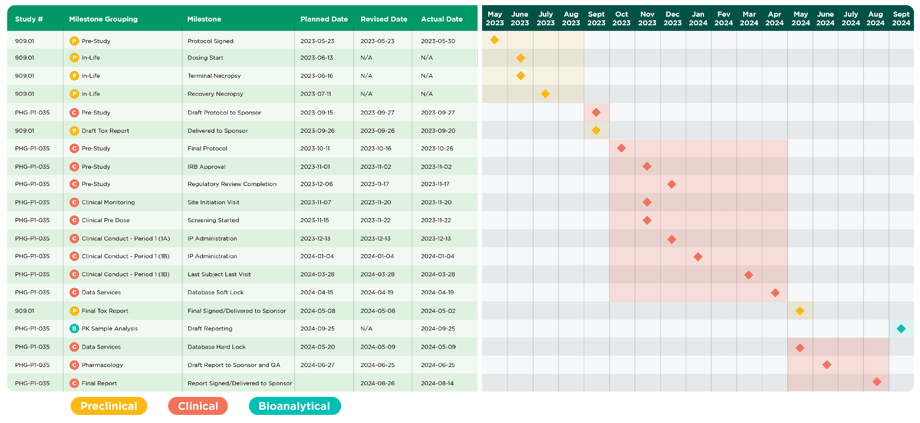
Case Study―Streamlining Clinical Trial Start-up for Accelerated Drug Development
Co-Located Laboratories
Our co-located bioanalytical laboratories provide real-time data access, enabling rapid decision-making, timely analysis, and quick PK data turnaround to support informed dosing decisions, ensuring your study remains both safe and effective.
Combining trial conduct and bioanalysis with Altasciences delivers significant benefits:
- Timed interim sample analysis for dose escalation decisions
- Rapid turnaround of PK/PD analysis between cohorts
- Biomarker assay development and validation for exploratory or primary endpoints
- On-site flow cytometry
- Centralized scheduling between clinic and lab
- Single point of contact with a dedicated program manager for streamlined communication
Learn more about our bioanalytical facilities.
Accelerated Phase I
Clinical Trials With Adaptive
Protocols and Real-Time Data
Altasciences’ specialized early-phase clinical trial services include adaptive cohort management and real-time contingency planning, allowing for responsive adjustments based on emerging safety data for efficient milestone achievement. With our proprietary scheduling system, Compass, we can provide real-time insights into key performance indicators, financials, project scope, milestones, deliverables, and quality control.
Our team works closely with you to keep each stage on track, ensuring a smooth startup and adherence to timelines.
How Adaptive Trial Designs Can Accelerate Early-Phase Drug Development
Our adaptive clinical trial designs and umbrella protocols enable optimized data collection to ensure maximum valuable information from a single clinical trial, and seamless progress in complex SAD/MAD studies. This allows for real-time adjustments based on data analysis, optimizing information for precise dose escalation, safety monitoring, and successful NDA submissions.
Adaptive, integrated designs enable multiple early-phase assessments to evaluate compound safety (imbedded cardiac safety, biomarker and AE management), absorption (pharmacokinetics [PK], food effect, ethnobridging, formulation bridging), interaction (DDI; drug-drug interaction), and early efficacy (biomarkers, patient arms).
A Turnkey First-in-Human Clinical Trial Solution
At Altasciences, our primary focus is on early-phase drug development, so your FIH trial will never compete for resources with larger, later-phase trials. We drive your molecules to clinical proof of concept, delivering end-to-end preclinical, clinical, bioanalytical, and manufacturing solutions to give your program the momentum it needs.
A one-stop solution, our early-phase services encompass everything from clinical trial design and protocol development to patient recruitment, data management, biostatistics, trial conduct, bioanalysis, and regulatory guidance.
Ready to start your first-in-human study?
Get in touch with us today!
Altasciences conducts your study with precision while providing full protocol development support. Seamless coordination between the clinic and bioanalytical lab ensures transparent communication and adherence to early-phase timelines.
Alongside our comprehensive core services for FIH trials, we offer in-house expertise for Phase I, available as FTE or per project, in:
- Study design & protocol development
- Medical monitoring
- Regulatory support
- Clinical trial monitoring
- Data management
- Biostatistics and programming
- PK/PD analysis
- Medical writing and reporting
Our processes maximize communication and efficiency, keeping your project on track. Centralized data storage ensures learnings carry through from preclinical research to manufacturing, clinical trials, and bioanalysis, with real-time access for all teams.
With unparalleled safety oversight and review procedures, we ensure all our operations and facilities are designed with participant safety as the primary objective in every trial—from initial feasibility and risk assessments through to study completion.
Our clinics’ layouts can be configured to the specific requirements of your program. This includes specialized areas for intense safety monitoring and procedures, secure facilities with Schedule I licensing, and in-house pharmacies with compounding and full sterile/non-sterile investigational product (IP) drug preparation capabilities.
- >30 trials completed per year
- Daily investigator assessment
- 24/7 Advanced Cardiac Life Support (ACLS) provider coverage on-site
- Basic Cardiac Life Support certification for all clinical staff
- Crash carts on-site
- Scenario-based response training
- Telemetry with pulse oximetry
- Strategically placed panic buttons
- Proximity to major hospitals
Experienced medical oversight and thoughtful communication with Medical Monitors and Safety Review Committees (SRC) provide robust and continuous safety monitoring.
Specialized Dose Escalation Evaluations
- PBMC collection and separation
- Biopsies
- CSF collection
- Asian/Non-Asian ethnobridging
Our clinic-ready, small molecule manufacturing experts quickly formulate your drug product under cGMP conditions and deliver it to the clinical site, ready for dosing. We offer a range of clinical trial materials, including liquid and powder-filled capsules, creams, gels, and more.
Seamless integration of processes between our CDMO and clinics, with shared methods and transparent data exchange, ensures your small molecule FIH trial kicks off on time and meets milestones throughout the program. If needed, certain dosage or formulation changes can be handled on the fly.
First-in-Human Resources
The Altascientist
First-in-Human Trials — FAQs
What are first-in-human (FIH) trials?
The U.S. Food and Drug Administration (FDA) Phase I trial includes the first human exposure to an investigational drug (IND). Phase I or FIH trials are typically not large, usually involving from 20-80 subjects. The objective of a classical first-in-human trial is to determine the safe dose range for further clinical development.
What data is required to support the start of first-in-human trials?
Prior to starting first-in-human trials, you will be required to submit preclinical and other safety data to the local regulatory authorities to evaluate, to ensure that your drug product is safe for human testing. In the U.S., the Food and Drug Administration (FDA) is responsible for managing drug approvals, and in Europe the European Medicines Agency (EMA) is the regulating body. In Canada, a Clinical Trial Application (CTA) must be submitted to Health Canada, including preclinical and safety data, for review and approval before the trial begins.
Are there any complementary trials to consider when planning my first-in-human trial(s)?
You may want to conduct an early QT study, in order to avoid the need for a more detailed thorough QT trial later in development. Depending on your intended indication and signals from safety data, you may also want to consider studying use in special populations or renal/hepatically impaired individuals, and you may need human abuse potential or cognitive/driving impact assessments.
Will I have a dedicated team supporting my first-in-human study?
Yes. For every first-in-human study, we build a dedicated, cross-functional team that looks after every part of your trial—including coordination with any third-party vendors. We treat your program as our own, staying closely aligned with your goals, timelines, and what matters most to you.
We carefully schedule activities so that your safety and bioanalytical data are available in time for dose escalation meetings, ensuring all study decisions are grounded in real data. Using our proprietary scheduling system and custom-designed, information-sharing platforms, our teams are always aligned around your goals and critical milestones.
Are you able to recruit the target populations needed for any first-in-human trial?
Yes. Altasciences has a database of 400,000 participants, with full medical screening profiles to allow for targeted outreach and recruitment efforts. We also have collaborative arrangements with local clinics and hospitals for studies involving patients.





