What Is Altasciences?
Altasciences is a forward-thinking, mid-size contract research organization offering pharmaceutical and biotechnology companies a proven, flexible approach to preclinical and clinical pharmacology studies, including formulation, manufacturing, and analytical services.
A Message From Our CEO
After more than 14 incredible years with Altasciences, I’m truly honored to step into the role of Chief Executive Officer.
I’ve had the opportunity to help build and oversee our diverse integrated capabilities, and through that experience, I’ve seen firsthand the passion, dedication, and scientific excellence that define who we are. Altasciences is more than an organization; it’s a community of talented people who care deeply about our clients, and each other. I’m incredibly proud of what we’ve built together.
We will continue to deliver what makes Altasciences different: an accelerated drug development platform that integrates our CRO and CDMO services, strengthens partnership, drives innovation, and keeps people at the center of everything we do.
Warm regards, Marie-Hélène Raigneau CEO Altasciences

Dare to Imagine With Altasciences
Imagine partnering with an integrated team that you trust, and telling your story once. Imagine a world where everyone and every project is of equal importance. Just imagine…
Our Story
For over 30 years, Altasciences has been partnering with sponsors to help support educated, faster, and more complete early drug development decisions.
Our integrated, full-service solutions include preclinical safety testing, clinical pharmacology and proof of concept, bioanalysis, manufacturing, program management, medical writing, biostatistics, clinical monitoring, and data management, all customizable to specific sponsor requirements.
Our shared goal: to help you make educated, faster and more complete early phase drug development decisions — to speed you to proof of concept and beyond.
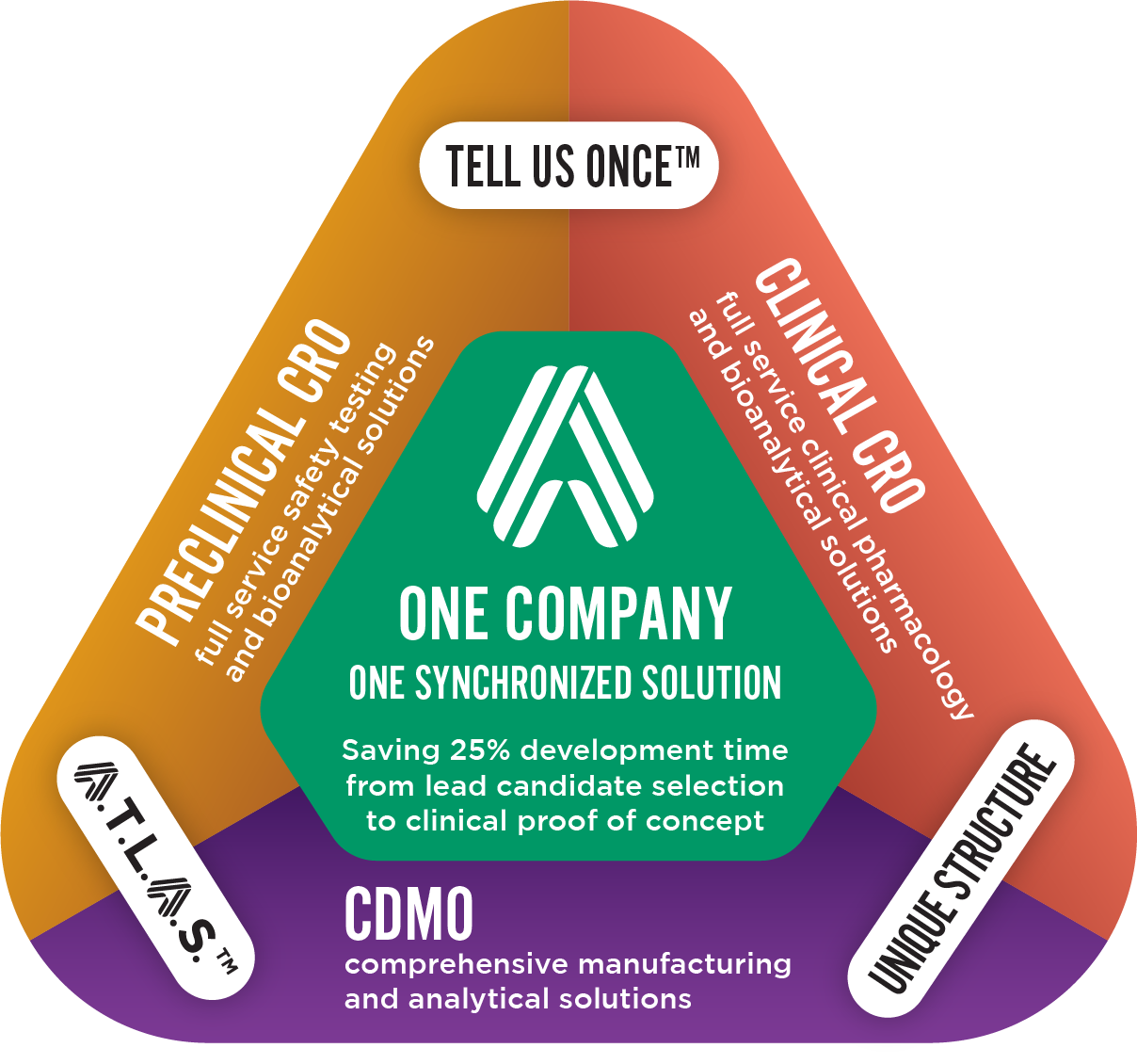
WHY PARTNER WITH USOUR UNIQUE APPROACH
At Altasciences, we provide you with clear, customized roadmaps, supported by our real-time data generation, proprietary communication platform, and central program management and scheduling to help you get to clinical proof of concept, faster.
Our Purpose
Our purpose is to help you research and develop potentially life-saving therapeutics — and get you there faster, with integrated CRO/CDMO services, always moving in unison for maximum efficiency. Our commitment to quality, excellence, integrity, and respect help us deliver big impact with a personal touch.
Our Facilities

1200 Beaumont Avenue
Montreal, QC H3P 3P1
Canada 514 381-2546
Our Origins
OUR EXECUTIVE TEAM
Marie-Hélène Raigneau holds a bachelor’s degree in psychology from McGill University and an Executive MBA from the Université du Québec à Montréal. She has spent over 15 years in leadership roles within the drug development industry, at both preclinical and clinical research organizations. She began her career as a Business Development Manager at Future Electronics and, in 2004, joined Charles River Laboratories, where she eventually rose to the role of Global Director of Marketing.
Marie-Hélène joined Altasciences in 2012 as Executive Director of Business Development and Marketing. She was promoted to General Manager of Altasciences’ Montréal and Laval sites in 2015, and subsequently to Executive Vice President of Research Services in 2018. In 2020, she was appointed Co-Chief Operating Officer, where she oversaw Altasciences’ clinical and bioanalytical teams, formulation, development and manufacturing capabilities, and research support services—including data management, biostatistics, project management, and medical writing. In 2025, she was appointed President of Altasciences, and in February 2026 Marie-Hélène became CEO of Altasciences.
BA in Psychology, Exec MBA
Cathy joined us in 2010 with the mandate of transforming our commercial offering to cater to the expanding innovator market, and now oversees our commercial strategy, including M&A activity. She started her career at CTBR Bio-Research Inc. (now Charles River Laboratories) and served as Senior Site Director of its Montreal facility.
BA in Psychology, MBA
Dr. Setnik joined Altasciences in 2019. In her previous roles as Vice President of Scientific & Medical Affairs at INC Research/Syneos Health (Early Phase), Senior Director, Clinical Sciences (King Pharmaceuticals and Pfizer, Inc), and Research Scientist (formerly Ventana / Decisionline Clinical Research) she has accumulated impressive experience in early phase clinical trials, regulatory filing, lifecycle management, and strategic initiatives in business development and clinical drug development. Dr. Setnik is also an Adjunct Professor at the University of Toronto, Department of Pharmacology and Toxicology and the Managing Director lead of the Cross Company Abuse Liability Council and chair of the Clinical Pharmacology Community of the Drug Information Association (DIA). She is also an active member and speaker at various congresses including the College on Problems of Drug Dependence; and is actively engaged in many aspects of abuse potential assessment including development of patient reported outcome instruments and contributing to post-marketing surveillance studies.
PhD in Pharmacology and the Collaborative Program in Neuroscience
Dr. Morelli is a member of the College of Physicians of Québec, Canada, with over 30 years' medical experience and over 20 years' global clinical research background. He joined Altasciences in 2013, as a GI consultant and then as Medical Advisor, followed by Clinical PI and now Chief Medical Officer and Executive Vice President Medical Affairs. Prior to joining Altasciences, Dr. Morelli was the Global Medical Director at MDS Pharma for 10 years, overseeing five clinical sites in Canada, the U.S.A., and Ireland. Dr. Morelli is an Associate Professor of Medicine at McGill University Montreal. He also continues to be active staff at the McGill University Hospital Center. In addition, he is Director of Gastroenterology/GI Endoscopy at one of the McGill University affiliated hospitals. He has been lead Principal Investigator in numerous multi-national, multi-center clinical trials.
Steve oversees all of Altasciences' preclinical solutions and sites. He started as a toxicologist for Huntingdon Life Sciences. At CTBR Bio-Research (now Charles River Laboratories) in Montreal, he built the Safety Pharmacology Department, and then moved to China to establish the Charles River Shanghai preclinical facility, which became the first non-clinical facility in China to be certified as OECD GLP compliant. He joined WuXi AppTec in Suzhou, China, as VP of Preclinical Services, before coming to Altasciences in 2014.
BS in Biological Sciences, MS in Toxicology
Robert joined Altasciences in 2012, focusing initially on the growth of our bioanalysis business. Prior, he worked at Charles River Laboratories as a Regulatory Inspector, where he oversaw the Bioanalytical Preclinical segment and managed government audits. He also spearheaded the training and process development programs for the Quality Assurance departments in several site locations. When he joined Altasciences, he transitioned to Business Development, with responsibilities that included key global account oversight.
BS in Physiology, Graduate Diploma in Business Administration
David started his career in 2000 as a Quality Assurance Inspector at CTBR Bio-Research (now Charles River Laboratories). At Pharmascience, he developed and implemented a GxP quality system for both bioanalytical and clinical operations. David has been a member of the Board of Directors of the Canadian Chapter of the Society of Quality Assurance, and served as VP and then President. He joined Altasciences in 2012.
BS in Biology
Julie-Ann joined Altasciences in 2014 to lead business development for our Kansas City site’s operations. In 2016, she was promoted to Vice President, Business Development and in 2017, was appointed to her current position. She began her career in 1998 at Origenix Technologies, a biotech company in Montreal. In 2003, she moved to CTBR Bio-Research (now Charles River Laboratories) where she progressed to Head of Marketing. In 2008, she was seconded to China as Director of Business Development for Charles River Laboratories Shanghai, and then served as Head of Global Marketing for the Charles River Laboratories Preclinical Division. In 2011, she joined WuXi AppTec in Suzhou, China, as Executive Director of Business Development for Preclinical Services, managing staff in China and the U.S.
BS in Exercise Science (Kinesiology), MBA
Mariano started his career with Ernst & Young in 1991 before joining Phoenix International as Global Director of Finance where he completed eight acquisitions. Subsequently, he held a number of Chief Financial Officer positions with various companies, including Galileo Genomics, Neurochem, and GFI Solutions. Most recently, Mariano was the Chief Operating Officer and Chief Financial Officer for Klox Technologies. He joined Altasciences in 2018.
B. Commerce, Graduate Degree in Public Accountancy, CPA CA, CPA USA
Consult the detailed biographies of our expert team members, by department
AWARDS
Recognized by the industry, Altasciences has the leadership, scientific, and operational excellence you need to move your programs from lead candidate selection to clinical proof of concept, and beyond.
OUR CLIENTS
We partner with biotech and pharmaceutical companies of every size and spend, from startups to global leaders. Our client portfolio is deep, international, diverse — and working at the forefront of medical science.
Social Responsibility
Our people take pride in engaging in the communities where we work and live. Initiatives include Dare to Give breast cancer fundraising, our school supplies campaign, Holiday gift drives for kids, food and winter clothing drives, as well as other ways of giving back. We reflect our passion, commitment, and dedication in everything we do, from our community involvement to our work.
Committed to a sustainable future
Building a healthier future extends beyond the groundbreaking therapies we support. Altasciences is committed to operating sustainably through our comprehensive Environmental, Social, and Governance (ESG) program. We continuously strive to minimize our environmental footprint while championing social responsibility. This dedication is reflected in our participation in the Science Based Targets initiative (SBTi) and the UN Global Compact, solidifying our commitment to a sustainable future.
COMMITMENT TO PARTICIPANTS
We value the people who participate in our clinical trials and contribute to advancing medical science. We, in turn, provide a safe, comfortable, and caring study environment. We treat all our participants respectfully, with transparency and openness, at the initial recruiting and screening process, and throughout the trial period.
FOCUS ON ANIMAL WELFARE
We maintain an unwavering focus on the welfare of the laboratory animals in our care. Our entire preclinical staff is trained in laboratory animal care and focused on animal welfare and environmental enrichment—embracing compassion, sensitivity and adherence to regulatory guidelines.
AI in Drug Development
Artificial intelligence is more than a technological advancement—it's a strategic enabler. We’re committed to using AI responsibly and intentionally to elevate our people, empower our clients, and transform the speed and precision of decision-making in drug research.
Learn More
ABOUT US - FAQs
Who owns Altasciences?
Altasciences is owned by Novo Holdings A/S, a world-leading life science investor with a focus on long-term, sustainable value creation. Founded in 1992, Altasciences is a leading, fully-integrated, early drug development solutions platform, providing the pharma and biotech industries with a trusted partner for drug development, from preclinical safety testing through clinical proof-of-concept studies. In the past decade, the company has grown both organically and through acquisitions to have a significant presence in the preclinical safety testing, clinical pharmacology, bioanalytical, CRO services, and CDMO markets. Headquartered in Laval, Canada, Altasciences operates eight facilities in the U.S. and Canada and employs over 1,800 people.
What does Altasciences do?
Altasciences is a contract research organization that offers a flexible approach to early phase drug development. Its integrated, full-service solutions include preclinical safety testing, clinical pharmacology and proof of concept, bioanalysis, formulation, manufacturing and analytical services, program management, medical writing, biostatistics, data management, and more.
What is a CRO?
A contract research organization (CRO) provides support and guidance to pharmaceutical, biotechnology, biopharmaceutical, and medical device companies with the research and development of their products, including generic or novel drugs, in the form of research services outsourced on a contract basis. A CRO is responsible for planning, conducting, and managing contracted research for its clients. At Altasciences, CRO support includes preclinical and clinical research, bioanalysis, formulation, manufacturing and analytical services, medical writing, scientific and regulatory guidance, data management, program management, biostatistics, and more.
What is a CDMO?
A contract development manufacturing organization (CDMO) is a company that supports pharmaceutical, biotechnology, and biopharmaceutical companies with not only the manufacturing of a drug product, but also its development prior to manufacturing. CDMO services are supplied on an outsourced contract basis. At Altasciences, CDMO services include formulation, analysis, method development, clinical supply, and commercial manufacturing.








































