Medical Writing Expertise Is at the Heart of Your Regulatory Submission
Our team of experienced medical writers provides clinical, medical, and regulatory expertise to support every stage of drug development. We prepare documents that demonstrate safety and efficacy, always aligned with current regulatory guidelines.
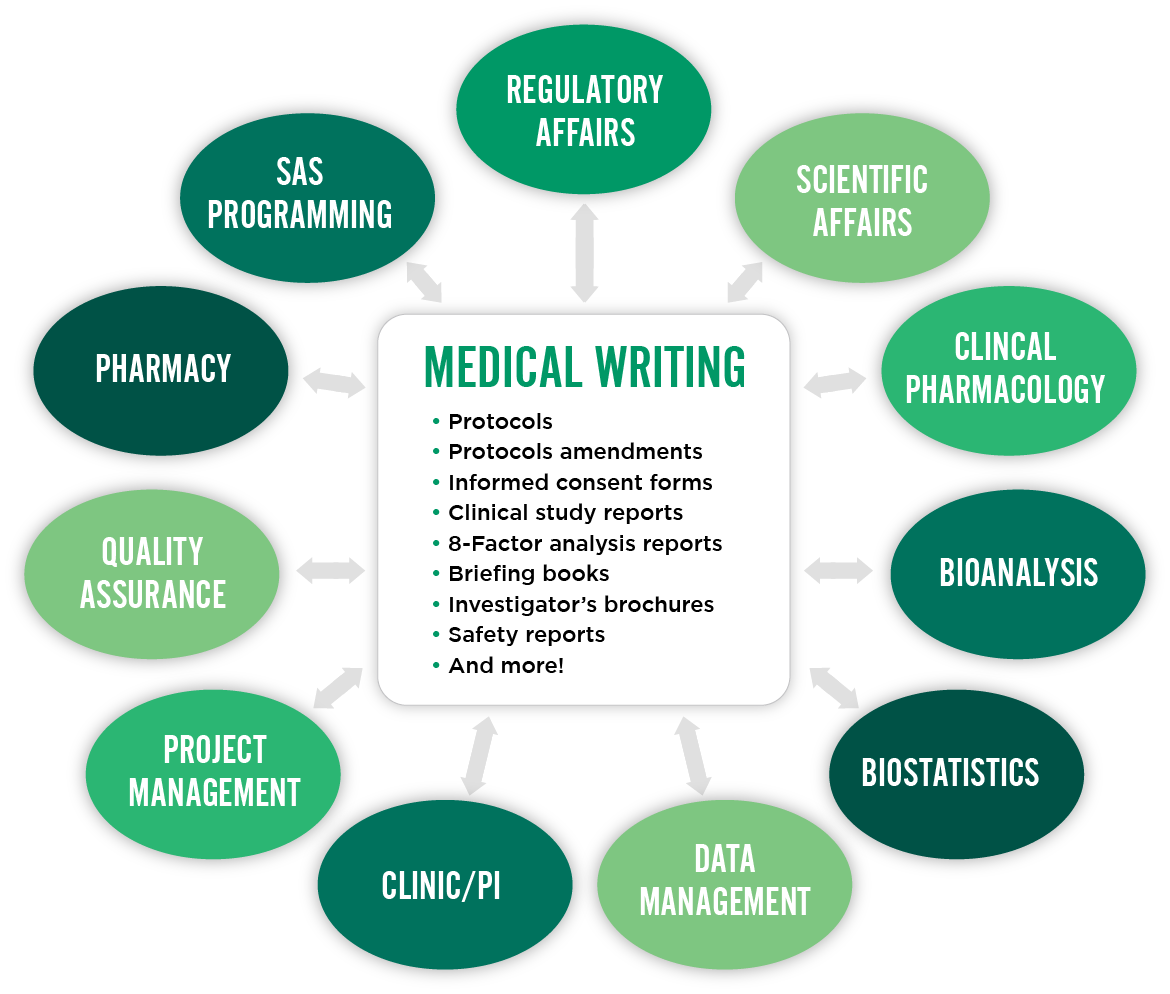
We work with clinical, medical, biostatistical, and bioanalytical experts to create accurate study protocols, informed consent forms, clinical study reports, and regulatory submissions. Our writers focus on clarity, accuracy, and compliance.
What we provide:
- protocol synopses
- protocols and amendments
- informed consent forms
- clinical study reports and addenda
- safety reports
- NDAs/BLAs marketing application documents
- investigator brochures
- briefing books
- annual reports/DSURs
Bringing Your Data to Life
Our medical writers have extensive experience covering a wide range of therapeutic areas, for small and large molecules. You can rely on our dedicated experts to design, prepare, write, proofread, edit, and format the full array of documents required for the smooth conduct of your clinical programs and facilitate submission of data worldwide.
Protocol Development
Our protocol-development scientists have expertise in pharmacology, pharmacokinetics, and a wide range of therapeutic areas. They use preclinical data to design studies from first-in-human through proof-of-concept. We provide protocols that meet regulatory and ethical requirements in North America, Europe, and Asia.
Clinical Trial Reporting
We collaborate with pharmacokinetic scientists, biostatisticians, investigators, and subject matter experts to prepare complete, accurate reports. Our writers link safety and pharmacodynamic data to assess proof-of-concept and safety throughout development. All reports are ICH-compliant and meet submission requirements.
Scientific Publications
We support manuscripts, posters, and abstracts with content development, review, and editing. Our writers bring scientific and regulatory expertise to ensure publications are accurate and well-structured.
Other Regulatory Documents
- Investigator Brochures: Summaries of investigational product data prepared to ICH standards, providing investigators and regulators with current, accurate information.
- 8-Factor Reports: Assessment of abuse potential using nonclinical and clinical data to inform regulatory decisions.
- Briefing Books: Concise, well-organized documents that present essential study information for regulators and stakeholders.
- IND Submissions: Compilation of clinical and nonclinical data into complete, compliant packages for regulatory review.
Experience
Our medical writing team has decades of experience across small and large molecules, multiple therapeutic areas, and global regulatory environments. We deliver documents that meet international standards, support efficient study conduct, and enable timely submissions.