Protocol Design Concepts in Phase I Ethnobridging Clinical Trials
In Phase I ethnobridging clinical trials, there are three primary protocol design concepts utilized. These concepts align the strategy with global clinical development and measure key factors necessary for meeting the unique needs of the Sponsor.
Pharmacokinetic/pharmacodynamic (PK/PD)
PK/PD modeling assesses the product’s sensitivity to intrinsic ethnic factors and is a common endpoint in ethnobridging trials. Intrinsic factors include race, height, weight, and other factors known to be affected by genetics. The reason that intrinsic (genetic) factors are measured instead of extrinsic (cultural or behavioral) factors is because intrinsic factors have a greater impact on the ability to extrapolate.
In PK/PD ethnobridging trials, there are specific drug properties that must be taken into consideration when designing the trial:
• Linear or nonlinear PK
• Concentration – Effect curve
• Therapeutic margin
• Metabolism
• Polymorphic
• Extensive
• Pro-drug requiring rapid conversion
• Protein binding
• Need for co-medications
• Likelihood for inappropriate use
Measuring receptor sensitivity through PK/PD modeling can lead to adjustments in dosage and other variables that define the study design. Monitoring these factors becomes increasingly important in ethnobridging trials where genetic variability may alter the exposure-response relationship.
Safety
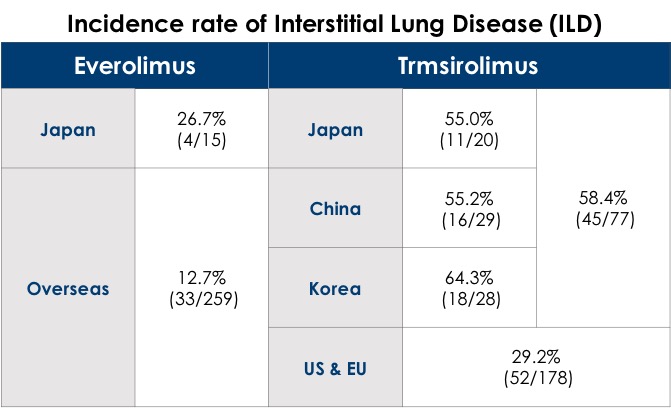
To ensure the safe testing of compounds in ethnobridging trials, it’s important to consider all of the studies that have previously been conducted with the investigational product, across all regions. This ensures that genetic differences in exposure-response relationships are taken into account when planning dosage and other facets of the trial. For example, in clinical trials of Temsirolimus and Everolimus, the adverse event (AE) incidence rate of Interstitial Lung Disease (ILD) in Asian populations was widely different than that of non-Asians1. As seen in the table below, the high incidence rate of this AE in Asians would be an essential element to consider when planning the ethnobridging trials for these products.
Anticipated Treatment Regimen
All of the factors above contribute to the treatment design in ethnobridging trials. Aligning the study to Global Clinical Development (GCD) is essential, requiring that dosage be aligned to later phase testing. This includes dosage amount, dosage timing, and other factors that could affect metabolization.
These three protocol design concepts shape the parameters of each individual ethnobridging study and allow the overall design to be tailored to specific drug properties. They highlight the importance of observing intrinsic factors in the development process, ensuring previous safety observations are taken into consideration, and aligning the study with global clinical development necessities. Overall, these concepts and observations help ensure that the duplication of studies is not necessary, which helps streamline global development.
Contact our experts to discuss your ethnobridging needs.
REFERENCE:
1. Oncology 2017;92:243-254; https://doi.org/10.1159/000457904



