Flow Cytometry Services
Our flow cytometry laboratory combines advanced technology with scientific expertise to deliver reliable, high-quality data for your preclinical and clinical programs. Equipped with state-of-the-art flow cytometers, we provide the precision and consistency you need to move confidently through each stage of development. From simple cell profiling to highly complex, multi-parameter studies, we tailor every solution to fit your program requirements.
What makes our approach different is our focus on flexibility and partnership. Every assay we develop and validate is designed with your goals in mind—whether you’re exploring an early discovery question, generating data for regulatory submissions, or evaluating immune responses in late-phase studies. Our team ensures that methods are both scientifically rigorous and regulatory compliant, so you can trust the insights you gain.
With the combination of cutting-edge instrumentation, scientific know-how, and a client-first approach, our flow cytometry services deliver more than just data—they deliver confidence. Partner with us to accelerate your program, overcome challenges, and bring your therapeutic innovations closer to patients.
Consult our Flow Cytometry fact sheet for details of our state-of-the-art equipment and capabilities.
 Regulated Services for Flow Cytometry
Regulated Services for Flow Cytometry
We develop and validate flow cytometry methods under GCP and GCLP and meet regulated study requirements from preclinical through clinical—covering primary endpoints and potency assays for product characterization.
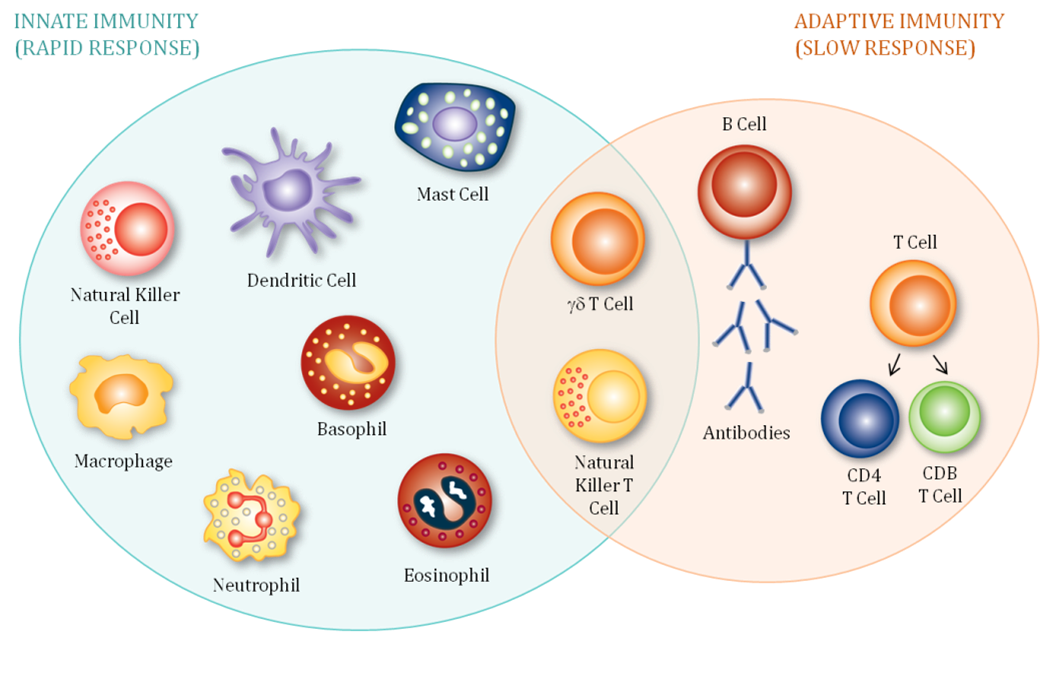
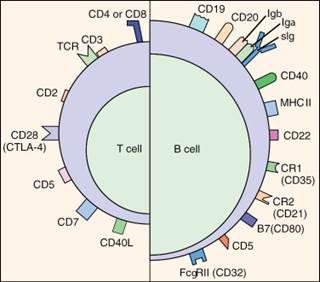
Dedicated flow cytometry core: immunophenotyping of leukocyte lineages and subsets, functional profiling, and cell enumeration.
Cell Lineage and Phenotyping
- T-cell subsets, thymocytes, B cells, plasma cells, NK cells, monocytes, dendritic cells, granulocytes, stem cells, platelets
- Activation Markers
- Intracellular cytokine stains
- NK cell killing assay (non-radioactive)
- Proliferation
- Reticulated Platelet Analysis
Enumeration
- Percentages x leukocyte counts from Advia 120
- BD TruCount tubes
Tissues
- Blood, spleen, lymph nodes, thymus, bone marrow
Advanced Flow Cytometry Services for Preclinical and Clinical Applications
If you wish to expedite the bioanalytical phase of your clinical or preclinical study or program, our flow cytometry services will help you get there. Our labs are located within a few minutes’ drive of our clinical pharmacology units, enabling rapid turnaround time between sample collection and analysis, as well as reduced costs. The close proximity between both laboratories can avoid potential risks at the primary endpoints of these types of trials, ensure rapid analysis and reporting of results which are necessary for the study objectives and ensuring seamless dose escalations for FIH trials.
| Preclinical Applications | Clinical Applications |
|
|
-
 Flow Cytometry Fact Sheet
Flow Cytometry Fact Sheet -
 Accelerate Your Data with our Co-Located Clinic and Lab
Accelerate Your Data with our Co-Located Clinic and Lab
LEARN MORE ABOUT OUR Laboratory SERVICES
Click below to explore more of Altasciences’ bioanalytical solutions.
Therapeutic Areas
Our deep expertise and capabilities in a broad range of therapeutic areas encompasses preclinical and early clinical studies for both small molecules and biologics. We can manage your entire program, as well as provide comprehensive support research services and bioanalytical expertise.