Making Early-Phase Drug Development Faster, Better, and More Efficient
Early-phase drug discovery and drug development are complex processes, where many moving parts can, and do, influence the success of a program. Managing multiple vendors for preclinical, manufacturing, bioanalysis, and early-phase clinical trials doesn’t need to be an additional challenge—engaging with a single partner who seamlessly and proactively advances your projects through the critical early stages, all the way through the regulatory approval process, and beyond, can make all the difference.
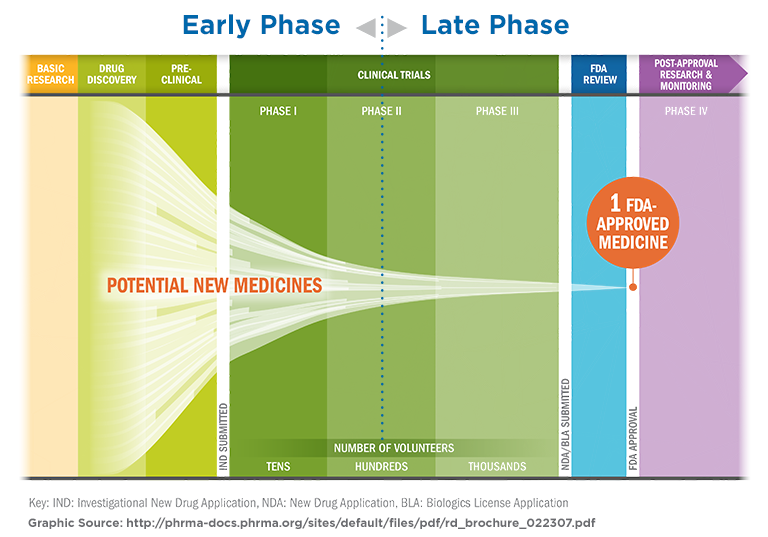
Given the high cost of drug development, and modest percentage of drugs that are safe and effective enough to make it to final market as an approved drug, maximizing the efficiency of programs for early-phase clinical trials is essential. Altasciences’ integrated, full‑service capabilities, supported by comprehensive research support services, can make the process seamless and efficient. Having a single‑source vendor reduces the administrative burden of multiple vendor agreements, reduces handoffs and the challenge of managing timelines across suppliers, simplifies communication and planning, and ultimately delivers a more synchronized experience.
The Benefits of Proactive Drug Development
At Altasciences, we champion Proactive Drug Development. This innovative approach aligns our decades of expertise around delivering a streamlined, synchronized early-phase drug development pathway, supported by specially designed systems, processes, and a unique organizational structure.
For your study, a dedicated, cross-functional program manager is your main point of contact. They use our proprietary scheduling system to manage your program timelines from API availability to final commercialization. Program management is supported by a fully transparent information sharing system (Tell Us Once™/Ask Albert), that ensures all key stakeholders have visibility on your studies, programs, and preferences, and adjust their work plans as your project advances.
As a full service CRO/CDMO vendor openly sharing information and proactively supporting decision-making, we are not limited by a linear, task-based approach. For example, we can update your Investigational Brochure while your Investigational New Drug (IND)‑enabling studies are in progress. The synchronization and communication allows us to plan your clinical studies while your preclinical safety assessment is ongoing, and you can start first‑in‑human (FIH) trials as soon as you receive regulatory approval for your drug application. We facilitate quick start-up of FIH clinical studies by timing small-scale drug formulations with clinical conduct, and make rapid, informed dosing decisions based on timed bioanalytical analysis and safety and efficacy data.
When project decisions are informed by concrete data, received in real-time, and analyzed by a knowledgeable, highly skilled team of experts, timelines can be reduced by up to 40%.
Structural Support for Smooth Integration
Altasciences fosters complete integration of all our services at a grassroots level, eliminating the internal silos that can impact your timelines. Our forward-thinking scientific experts become an extension of your team, dedicated to advancing your studies.
Our company is structured with all scientific teams reporting into two Co-chief Operating Officers, who ensure integration, harmonization, and collaboration across services, sites, and specialty areas to deliver the best value for you. We are aligned around a common objective: to deliver high-quality data for the studies and programs entrusted to us.
Additionally, our adaptive clinical trial designs allow sponsors to examine the data as it becomes available, and then adjust upcoming activities based on real-time results. In a standard dose escalation study, if early results show that doses need adjustment, or if additional cohorts of participants are required, the design could only be changed with an amendment, as well as regulatory and IRB approval.
Altasciences has compounding pharmacies in both the United States and Canada, that are integrated with our bioanalytical laboratories, designed to save you time. Our bioanalytical facility in Laval, QC, for example, is only a 15-minute drive away from our Montréal clinical pharmacology unit (CPU), which enables timely collection, processing, transport, and rapid analysis of your samples; and integration with our CDMO can also ensure clinical trial drug products are available on time.
Using integrated drug development services and clearly defined project support can accelerate your overall timeline, as our teams are completely integrated, and all projects are overseen by a dedicated cross-functional program manager. We maximize access to all the relevant data as it becomes available, and work to implement process efficiencies and mitigate delays.
References:
- Innovation in the pharmaceutical industry: New estimates of R&D costs. Link: https://www.sciencedirect.com/science/article/abs/pii/S0167629616000291
- A Tough Road: Cost To Develop One New Drug Is $2.6 Billion; Approval Rate for Drugs Entering Clinical Development is Less Than 12%



