JAK1-siRNA Treatment for Inflammatory Skin Diseases: Real-World Nonclinical Insights
Findings shared here were originally presented in a poster session at ACT 2025
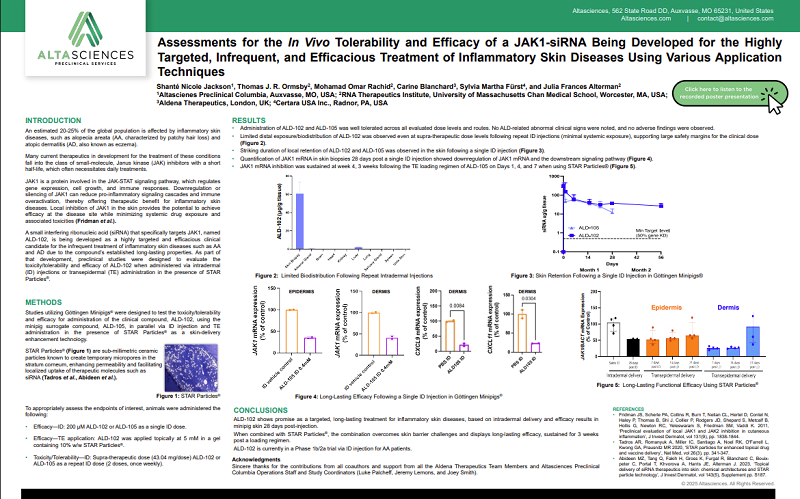
Inflammatory skin diseases, such as atopic dermatitis and alopecia areata, affect 20-25% of the global population, highlighting a significant unmet medical need. While Janus kinase (JAK) inhibitors have emerged as a promising therapeutic class, many are small molecules which require frequent dosing due to short half-lives. Recent nonclinical work by Shanté Nicole Jackson, Study Director and Senior Scientist at Altasciences, and colleagues , explored a novel approach: a JAK1-targeting small interfering RNA (siRNA) designed for localized, long-lasting efficacy with reduced systemic exposure.
JAK1-siRNA in Nonclinical Skin Disease Research
The studies evaluated ALD-102, a clinical compound siRNA targeting JAK1 (a key regulator of inflammatory signaling pathways) in parallel with the minipig surrogate compound (ALD 105) to ensure pharmacologic activity in the test system for adequate assessments. By downregulating JAK1 locally in the skin, this approach aimed to reduce inflammation while minimizing systemic toxicity
To support translational relevance, the team used miniature swine (strain: Göttingen Minipigs®), a well-established nonclinical model for dermatologic research due to the anatomic similarity to human skin (including dermal thickness, subcutaneous tissues, and underlying muscular structures). The studies explored two delivery approaches: intradermal (ID) injection and transepidermal (TE) application in the presence of STAR Particles®, sub-millimetric ceramic particles known to create temporary micropores in the stratum corneum.
Intradermal and Topical Delivery Approaches
Nonclinical evaluations were designed to assess both efficacy and tolerability across ID and TE delivery approaches. For ID administration, a single dose was used to evaluate efficacy, while repeat dosing at supra-therapeutic levels was conducted to assess tolerability. Additionally, TE delivery was performed using a topical gel formulation of the JAK1-siRNA in combination with STAR Particles®, enabling enhanced skin penetration.
Endpoints focused on characterizing local skin retention, systemic exposure, and pharmacodynamic effects, including changes in target gene (JAK1) mRNA expression in skin tissue over time.
Results: Sustained JAK1 Inhibition with Favorable Tolerability
Across all evaluated dose levels and delivery routes, ALD-102 and the minipig surrogate (ALD-105) demonstrated a favorable tolerability profile, with no treatment-related clinical signs or adverse findings observed.
Even under supra-therapeutic conditions, systemic exposure remained minimal, indicating limited biodistribution beyond the site of administration and supporting a strong safety margin.
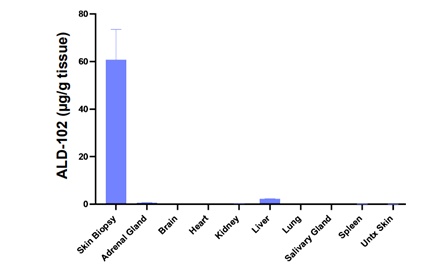
Following intradermal dosing, both compounds showed prolonged retention within the skin, highlighting the potential for sustained local activity.
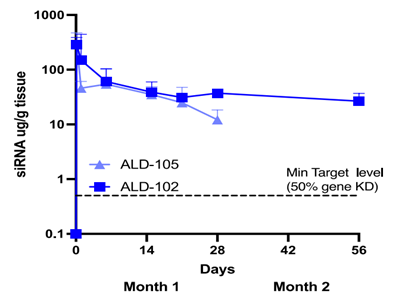
This was further supported by pharmacodynamic assessments, which demonstrated significant downregulation of JAK1 mRNA and downstream signaling pathways in skin tissue up to 28 days after a single dose.
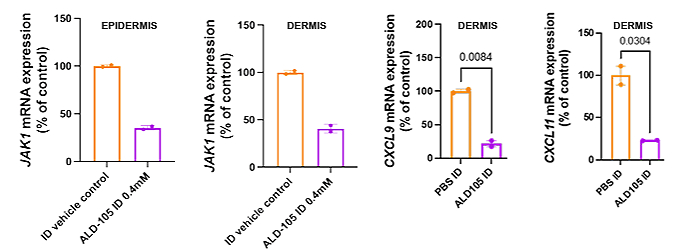
Transepidermal delivery using STAR Particles® also achieved meaningful target engagement. After a short loading regimen (3 doses over 7 days), JAK1 inhibition was maintained for up to 3 weeks, indicating that enhanced topical delivery can produce durable biological effects comparable to intradermal administration.
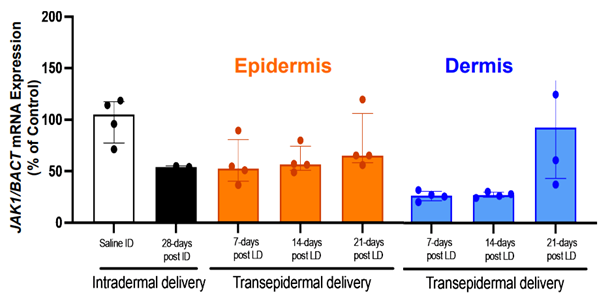
What This Means for Skin Disease Drug Development
These results highlight several advantages for drug developers pursuing JAK1-targeted therapies:
- Potential for infrequent dosing due to long-lasting activity;
- localized efficacy with reduced systemic exposure and toxicity risk;
- flexible delivery approaches, including non-invasive topical administration; and
- strong translational relevance supported by minipig nonclinical models.
Together, these attributes can help reduce development risk, improve patient compliance, and differentiate therapies in a competitive dermatology landscape.
Conclusion: Advancing JAK1-Targeted Therapies
These studies demonstrated that ALD-102 has the potential to deliver targeted, durable therapeutic effects in inflammatory skin diseases. Both ID and TE approaches showed promising tolerability and sustained JAK1 inhibition in minipigs, supporting further and ongoing clinical development. For a more detailed view of the study design and data, see the full scientific poster that was presented at the 2025 annual meeting for the American College of Toxicology.
For clients, these findings reinforce the value of integrating advanced delivery technologies, predictive nonclinical models, and molecular targeting strategies early in development to optimize outcomes and streamline translation to the clinic.
Altasciences offers nonclinical services that leverage minipig models, and advanced delivery systems for innovative therapeutics such as siRNA-targeted JAK1 inhibition. Get in touch today to learn how our team can help advance your next dermatology program from early discovery through clinical readiness.
Acknowledgements
Altasciences expresses its thanks for the contributions from all coauthors of the poster, “Assessments for the In Vivo Tolerability and Efficacy of a JAK1-siRNA Being Developed for the Highly Targeted, Infrequent, and Efficacious Treatment of Inflammatory Skin Diseases Using Various Techniques”, and support from the team at Aldena Therapeutics.
Thomas J. R. Ormsby, Mohamad Omar Rachid, Carine Blanchard, Sylvia Martha Fürst, and Julia Frances Alterman | Altasciences; RNA Therapeutics Institute, University of Massachusetts Chan Medical School; Aldena Therapeutics; Certara USA Inc.
FAQ: Intradermal and Topical Dermatology Studies
What is JAK1?
JAK1 is a protein involved in the JAK-STAT signaling pathway that regulates gene expression, cell growth, and immune responses. Downregulation or silencing of JAK1 can reduce pro-inflammatory signaling cascades and immune overactivation, thereby offering therapeutic benefit for inflammatory skin diseases.
How does intradermal delivery improve the safety profile of JAK1-targeted siRNA for skin diseases?
Intradermal administration delivers the JAK1-targeted siRNA directly into the dermis, the primary site of inflammation in many skin diseases. This localized approach ensures high drug concentration at the target site while maintaining minimal systemic exposure.
In nonclinical evaluations, this delivery method demonstrated a favorable safety profile and a strong safety margin, even at supra-therapeutic doses, by reducing the risk of off-target systemic toxicities.
Why are minipigs an appropriate animal model for dermatology studies?
Minipigs have skin that closely resembles human skin in terms of thickness, structure, and barrier function. This makes swine highly predictive for evaluating dermal absorption, local tolerability, and pharmacodynamic responses. As a result, this animal model is well suited for assessing dermal delivery of compounds targeting skin diseases. The translational relevance helps sponsors de-risk the transition from laboratory research to human clinical trials.
What are the benefits of using STAR Particles® for transepidermal delivery in dermatology?
STAR Particles® offer a non-invasive, topical alternative to intradermal injections by enhancing the skin penetration of large molecules like JAK1-siRNA. This technology allows the therapeutic to bypass the stratum corneum barrier effectively. In the nonclinical minipig studies discussed, this delivery method achieved meaningful target engagement and sustained gene silencing, offering a flexible and patient-friendly dosing option for chronic inflammatory skin diseases.
How long does JAK1 mRNA downregulation last after a single intradermal dose?
Nonclinical data in the minipig studies discussed indicate that a single intradermal dose of JAK1-siRNA can lead to significant downregulation of mRNA expression for up to 28 days. This prolonged local retention and sustained efficacy suggests the potential for infrequent dosing regimens in clinical settings, which could significantly improve patient compliance for those suffering from chronic inflammatory skin diseases.
This article was first published March, 2026.



