Clinical Trials
Shaping the Future of Microsampling at Altasciences by Jeff Plomley
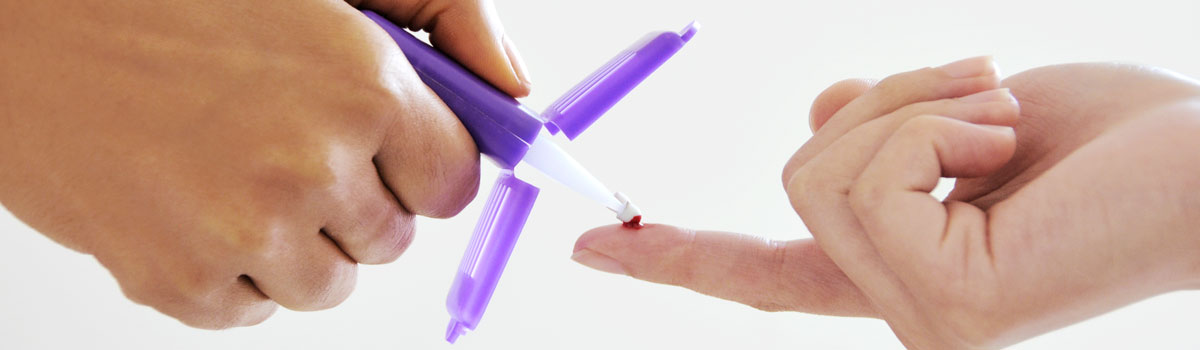
In recent years, healthcare has been undergoing a subtle yet profound transformation.
Navigating to the Clinic: Key Aspects of Translational Pharmacology, By Bruce Morimoto
Drug development is not for the faint of heart; a twist on the Hunger Games saying, “May the odds be (n)ever in your favor,” might adequately characterizes the challenge. As scientists, however, we manage these inherent risks by establishing best practices and learning from our collective successes and failures, as well as. In this field, experience truly matters.
First-in-Human-Enabling Toxicology Programs: Common Misconceptions by Grace M. Furman
Nomination of a clinical candidate and subsequent conduct of toxicology studies to support a first-in-human (FIH) clinical trial is an exciting milestone for sponsors! While many are highly experienced drug discovery professionals, some may have limited, or even no experience, in early clinical development, including the critical toxicology studies required to successfully support a FIH trial.
How Clinic-Ready Manufacturing and Adaptive Clinical Trials Can Reduce Drug Development Timelines
The complexities of active pharmaceutical ingredient (API) formulation, product testing, and documentation requirements can hinder the speed at which a promising drug candidate progresses to and through clinical trials.
The Year in Review 2025
Case Study: Applying Scientific Expertise to Optimize Complex Safety-Focused DDI Studies
Drug-drug interactions (DDIs) can pose significant safety risks, particularly in cardiovascular therapies, where patients are often on multiple medications. By analyzing these interactions early, we can help your studies stay on track and avoid potential regulatory roadblocks.
Case Study—Accelerating Recruitment in Renal Impairment Trials: Leveraging Specialized External Sites
STAT: Responsible AI Frees Scientists to Focus on Science
CLINDMO: Where Clinical Innovation Meets Manufacturing Excellence