Shaping the Future of Microsampling at Altasciences by Jeff Plomley
In recent years, healthcare has been undergoing a subtle yet profound transformation. Advances in digital health, telemedicine, and decentralized clinical trials are gradually shifting care away from traditional clinical environments and into patients’ daily lives. At the center of this shift is a growing emphasis on patient-centric approaches that prioritize accessibility, convenience, and real-world data collection.
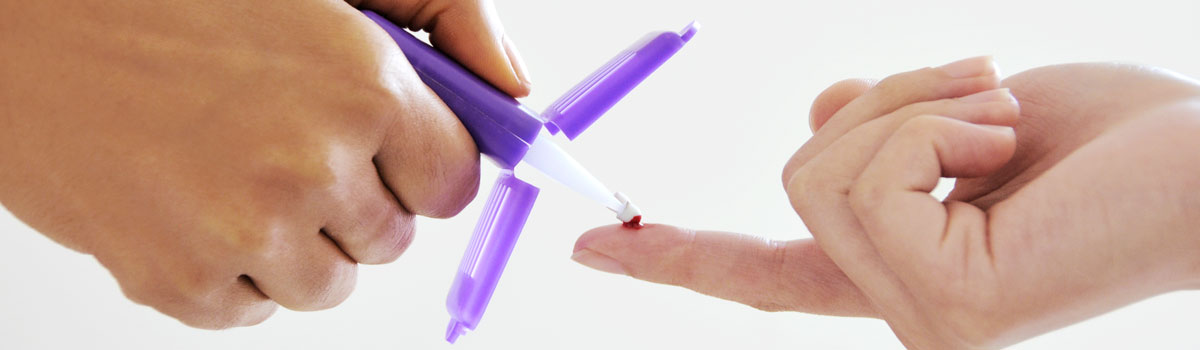
One technology at the heart of this movement is blood microsampling, a patient-centric method that enables individuals to collect small, accurate, and precise amounts of blood, typically 10 – 50 µL, outside of a clinical laboratory, often from home. For pediatric, elderly, or otherwise vulnerable populations, it provides a less invasive alternative to venipuncture, reducing discomfort, anxiety, and risks associated with poor venous access. It can also ease the burden for individuals with needle phobia.
From an operational perspective, microsampling simplifies workflows. By eliminating the need for plasma separation, it accelerates sample processing and shortens clinic visits, improving both patient experience and site efficiency. Smaller sample volumes also reduce logistical complexity, making handling, storage, and shipping more manageable. Additionally, microsampling can be seamlessly integrated into routine visits, enabling pharmacokinetic (PK), therapeutic drug monitoring (TDM), and biomarker assessments without adding procedural burden, ultimately enriching clinical trial data.
While venipuncture remains the gold standard, it comes with inherent challenges, including the need for trained personnel, scheduled visits, travel, and some patient discomfort. These barriers are particularly significant for individuals in rural areas, those with limited mobility, and patients requiring frequent sampling. Microsampling offers a compelling alternative: a minimally invasive, accessible method for collecting high-quality blood samples with minimal disruption to daily life.
Why Patient-Centric Microsampling Matters in Drug Development?
Improved Drug Efficacy Correlation
Remote microsampling enables blood collection during clinically relevant events, such as seizures or migraines. Capturing drug concentrations at these critical moments strengthens the correlation between exposure and therapeutic effect, providing more meaningful efficacy data.
Data Enrichment Through Multidimensional Measurement
Low-volume sampling enables more frequent collection without increasing patient burden. This higher sampling density supports richer pharmacokinetic and pharmacodynamic (PK/PD) profiles, which are particularly valuable in early-phase trials, pediatrics, rare diseases, oncology, infectious diseases, and advanced therapies. More time-points lead to more accurate parameter estimation.
Beyond drug concentrations, microsamples can support biomarker, metabolomic, proteomic, genomic, and epigenetic analyses. This enables integrated, longitudinal datasets derived from minimal blood volume, facilitating more comprehensive insights into disease and treatment response.
Enhanced Population Pharmacokinetics and Modeling
Microsampling aligns well with population PK (PopPK), physiologically based PK (PBPK), and Bayesian modeling approaches. Frequent, low-volume sampling across diverse patient populations improves the characterization of inter-individual variability and supports robust covariate analysis, including factors such as age, weight, and genetics.
Enabling Clinical Research Through Microsampling Device Technologies
Technological innovation has played a pivotal role in advancing microsampling. The introduction of the Mitra® Volumetric Adsorptive Microsampling (VAMS®) device by Neoteryx® in 2015 marked a significant milestone by enabling accurate, reproducible collection of finger-stick blood volumes while eliminating hematocrit-induced sampling bias, an issue that previously limited broader adoption in regulated bioanalysis (Fig 1).
Shortly thereafter, Tasso introduced on-body collection devices that combine microneedle-based sampling with vacuum-assisted blood collection. The newest version of the device, the Tasso T-20, provides a nearly painless, user-friendly experience and further expands the feasibility of at-home sampling, all with push-button ease.
Both the Mitra® and Tasso devices use a rigid porous polymer (RPP) substrate for blood absorption, and while this allows accurate and precise volumetric sampling, RPP’s can introduce other bioanalytical challenges. For example, with an inadequate sample preparation workflow, recovery can decrease with increasing blood hematocrit (HCT) due to analyte trapped by erythrocytes in the pores of the substrate. Further, analyte yields can decrease over time due to alterations in dried blood morphology. Such “age-related extractability” phenomena can be misinterpreted for analyte instability.
To mitigate these challenges, the LC-MS R&D team at Altasciences innovated a workflow which leverages controlled compression and decompression of the substrate during solvent extraction. The strategy was to treat RPPs as hard sponges that could absorb extraction solvent and then be “wrung out” by compression using impact with a bead, mechanically disrupting erythrocyte-related analyte trapping (Fig 2).
The compression/decompression cycling of the substrate (like the soaking and wringing of a sponge) overcomes the deleterious consequences caused by HCT and the workflow was coined “Impact-Assisted Extraction”, with initial results first published in the journal Bioanalysis [Vol 9 (22): 1761-1769 (2017). Since then, the impact-assisted extraction workflow has been applied to numerous small and large molecule applications (e.g. antisense oligonucleotides, monoclonal antibodies) for a diverse range of clinical indications (e.g. epilepsy, migraines) in a variety of study populations, including pediatrics, with microsamples collected either in-clinic or at-home.

An alternative means to overcome RPP challenges is to use cellulose as blood collection substrate since there is reduced analyte trapping by erythrocytes and therefore less aggressive universal workflows may be applied for extraction (e.g. sonication). Specifically, the Capitainer®B microsampling device is particularly well suited for quantitative bioanalysis since a volumetric aliquot of blood is transferred from a dedicated sampling inlet to a pre-cut Ahlstrom grade 222 cellulose disc via an exact volume microcapillary channel (Fig 3), with options available for 10 µL or 50 µL of collected fingerstick blood.
Available in a convenient card format, the engineering design has recently been applied to the generation of dried plasma spots (DPS) from a blood sample in the SEP10 product by incorporation of a HCT-removal membrane. Altasciences is currently examining the applicability of DPS technology to overcome HCT-induced analyte instability when that becomes a dried blood challenge. Stay tuned!

Overcoming Stability Challenges in Clinical Microsampling
Analyte stability in dried blood samples remains an important consideration. One solution to confer stability is to ship and store samples at reduced temperatures. However, this undermines a primary advantage of microsampling, and that is, the ability to ship samples at ambient temperature—a capability that facilitates remote collection.
A more effective solution involves pre-treating collection substrate with stabilizing chemistries that prevent analyte degradation, as this approach preserves sample integrity while maintaining the practicality of remote collection. In an industry first, Altasciences recently developed and validated a Mitra® assay using this strategy, with sampling kits prepared in-house and with successful correlation of the pre-treated substrate shelf-life with conferred microsample stability.
Recently, in collaboration with GatesMRI for the development of a microsampling assay to support an anti-tuberculosis (TB) drug panel, an antioxidant was applied to the Capitainer®B substrate to stabilize bedaquiline and its metabolite, N-desmethyl-bedaquiline. Until now, these analytes had been notoriously unstable in dried blood assays, limiting the utility of the technique for this class of compounds. However, by identifying degradation pathways in aged microsamples by LC-MS/MS and correlating their elevated expression level with increased HCT, an optimized stabilizing chemistry could be applied prior to blood collection, resulting in up to 68 days of stability. The novel anti-TB workflow leveraging pre-treated Capitainer®B devices was recently presented in a Patient-Centric Sampling Interested Group (PCSIG) webinar and presented at the 2026 MSACL conference.
Looking Ahead
Microsampling continues to gain momentum as a transformative tool in clinical research and drug development. Ongoing innovation in device design, improved downstream analytical sensitivity and overcoming logistical hurdles are expected to further expand its applicability and reliability.
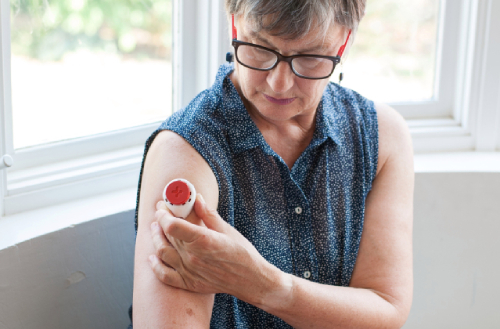
the Tasso microsampling device.
For example, Capitainer®’s next generation prototype device integrates electronics powered by a bio enzymatic fuel cell that allows capture of sampling time and the recording of temperature and humidity during shipping, significantly aiding remote collection.
As industry continues to prioritize patient-centric approaches, microsampling is poised to play an increasingly central role in enabling more flexible, efficient, and data-rich clinical trials. The recent Off-Label With Altasciences podcast featured experts from Pfizer, GatesMRI, and the PCSIG, and highlights emerging trends and innovations that point toward continued growth and broader adoption of these technologies.
Microsampling at Altasciences
With over fifteen years of clinical and bioanalytical expertise in microsampling collection and analysis, Altasciences provides comprehensive end-to-end solutions in support of drug development, including: microsample kit preparation, advanced workflows carefully optimized for the extraction of both small and large molecules from dried blood samples, and the establishment of blood-plasma concordance in support of bridging studies.
Looking to incorporate microsampling into your clinical study? Get in touch today to learn more.
Want to learn more about microsampling? You might be interested in the resources below:
- Podcast: Microsampling—When Small Steps Lead to Big Outcomes
- Podcast: Sparking the Evolution of Microsampling with the Mitra® Vams® Device
- The Altascientist Issue 16: Microsampling in Drug Development
- Video: Altasciences’ Bioanalysis Services—Microsampling
- Educational video: A Strategic Approach to Microsampling Device Selection and Associated Workflows Supporting Regulated Bioanalysis
About the Author: Jeff Plomley

Jeff Plomley joined Altasciences in 2016 having begun his research career in the Gas Phase Ion Chemistry Laboratory of Professor Raymond E. March designing novel ion trap scan functions. He then joined Thermo Instruments Canada as an applications chemist in mass spectrometry and later SCIEX in core research. Jeff has worked in the CRO environment since 2001, developing over 250 de novo LC-MS/MS assays supporting drug development in both preclinical and clinical applications.
Jeff has contributed to the publication of over 25 peer-reviewed papers and numerous scientific posters and technical publications, holds patents on MS instrumentation, and frequently presents on microsampling workflows and advanced MS techniques.
Jeff’s current research interests include the implementation of novel workflows and applications leveraging microsampling technology.
Connect with Jeff on LinkedIn.
FAQ: Microsampling in Drug Development and Clinical Trials
What is microsampling in blood testing?
Microsampling is a minimally invasive blood testing method that collects small volumes, often via finger-stick. It enables remote sample collection in clinical trials, supporting decentralized studies and improving pharmacokinetics data capture.
What are the key benefits of microsampling for drug development?
Microsampling allows more frequent sampling with less burden, generating richer pharmacokinetic data. This improves drug exposure analysis, supports advanced modeling, and enhances decision-making in clinical trials.
Why is microsampling better for patients in clinical trials?
Microsampling reduces clinic visits, discomfort, and logistical complexity. It’s especially useful for remote, pediatric, or elderly populations, improving participation and overall clinical trial efficiency.



